I Heart Phenolics
While getting my hair colored for the holidays, my hairdresser recounted the itinerary from his recent South African getaway. His trip included a stay in Stellenbosch, a famous wine growing region, where he lustily overindulged in the local red wines, which he “absolutely cannot” tolerate here “because of sulfites.” He went on to explain that South African wines contain fewer sulfites allowing one to drink all day long, without feeling bad. “What do you drink when you’re at home?” I asked.
“Chardonnay,” he quipped with flourish.
Now I have no idea whether or not South African wine producers employ a lower concentration of sulfites than we Californians do, but it is an interesting question. More pointedly, though, I can definitively tell you that the preservation of white wine requires more sulfite than red ones. So drinking white wine domestically but red vino abroad exemplifies the miraculous, phantasmagorical power of what I call The Vacation High. Drinking anything in South Africa’s wine country sounds divine but fails to unravel the myth of sulfites. So why is it that red wine requires less SO4 than whites? It’s the phenolics, the lovable chemical compounds with the funny name. And they look like this:
************DISCLAIMER- CARTOONS ARE MERELY A USEFUL PICTORAL- TEXT TO FOLLOW IS ENTIRELY COMPREHENSIBLE.********************
(Sorry, guys. I know I’m a real S.O.B to lure you with a sassy hairdresser anecdote only to bait-and-switch with the biochemistry…)
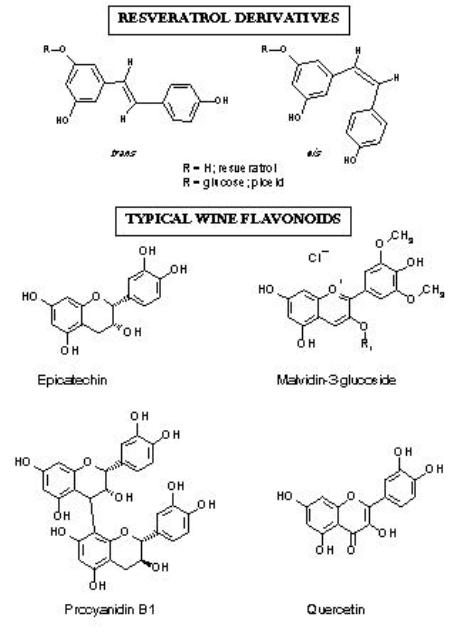
Phenolics, sometimes referred to as polyphenols, are chemical compounds produced by plants, including grapevines. The phenolics we care about are soluble chemical compounds located predominantly in grape skins and seeds. Phenolics include things like tannins, which make your mouth feel dry and puckery and the anthocyanins that color red wine red. As you know, grape juice from both red and white grapes is clear. Red wines are hued because they are fermented in conjunction with the grape skins; white wines are not. In other words, to make white wine, you smoosh grapes and ferment only the juice, discarding the skins, pulp, seeds, and stems. With red wines, you squash the grapes and mix the juice, pulp, skin, seeds, and maybe even some stems together and then ferment the whole thick, gooey glob en masse. Then you drain off the juice later. This means that through the process of fermentation, unique skin and seed components are extracted into red wine that are absent from whites. These diverse compounds are united in that their chemical silhouette each includes a hexagon-shaped ring. Beyond that, the compounds look and function differently, modifying different aspects of a wine’s personality, taste and mouthfeel.
There are 6 different classes of soluble phenols. Only one team, the cinnamate esters, is found in the pulp, their great distinction being the only soluble phenol present in white wines. Two different phenolic gangs control color: the anthocyanins and the flavonols. Players on tribe anthocyanin have names like peonidin, delphinidin, and petunidin-3-glucoside. Of course these compounds were first isolated from a colorful garden of peonies, delphinium, and petunias before being noted in grape skins too. The flavonols are color co-factors that make red wines appear richer and redder. The flavan-3-ols live only in grape seeds and taste bitter. This is why we squeeze the grape skins and seeds so judiciously at press, lest we crack the seeds and leach the bitterness into our finished product. When the flavan-3-ols congregate into chains called polymers, they make tannins. Tannins, of course, are responsible for astringency. Then over time, as wines age, the tannin chains grow even longer, softening that distinct, mouth-puckering quality. Tannins also polymerize with oxygen exposure, via oxidation reactions. In a way, tannins act like an oxygen sponge, absorbing the harmful effects of oxygen without wrecking the juice. Since red wines contain more phenolics than whites, they can absorb, or “consume” more oxygen without detrimental effect. In fact, sometimes oxygen exposure improves red wines, by mimicking and hastening the effects of aging thereby mellowing any acerbic, tannic harshness. In contrast, white wines fade from vibrant straw and honeyed hues to murky brown after very little exposure to ambient air. This is why white wines require more sulfites, potent anti-oxidants, to maintain their delicate color. Hearty red wines already posses a built in oxygen buffer through the phenolics extracted from the grape skins and seeds. (Plus brown discoloration is more obvious in pale, white wines than inky, purple reds).
Lastly, phenolics are important to our health. You have probably heard about them on 60 Minutes or read about them in the newspaper. Indeed many of the cardio-protective effects of red wine are attributed to phenolics, in particular the final chemical class called “stilbenes.” Within squad stilbene, the most famous player is resveratrol, touted to reduce heart disease, prevent dementia and diabetes, protect against colds and influenza, increase bone density, and even slow aging. As you can imagine, resveratrol is the focus of frenzied scientific and drug research. But before you guzzle away your inhibitions in the name of science and good health, remember “a 150 pound man would have to drink 1,500 bottles of pinot noir a day to get the same dose of resveratrol that [one researcher] gave his mice.” (Wine Spectator, May 31, 2009). So yes, science supports red wine for healthy hearts because red wines are chalk full of phenolics, the loveable chemical compound with the funny name.